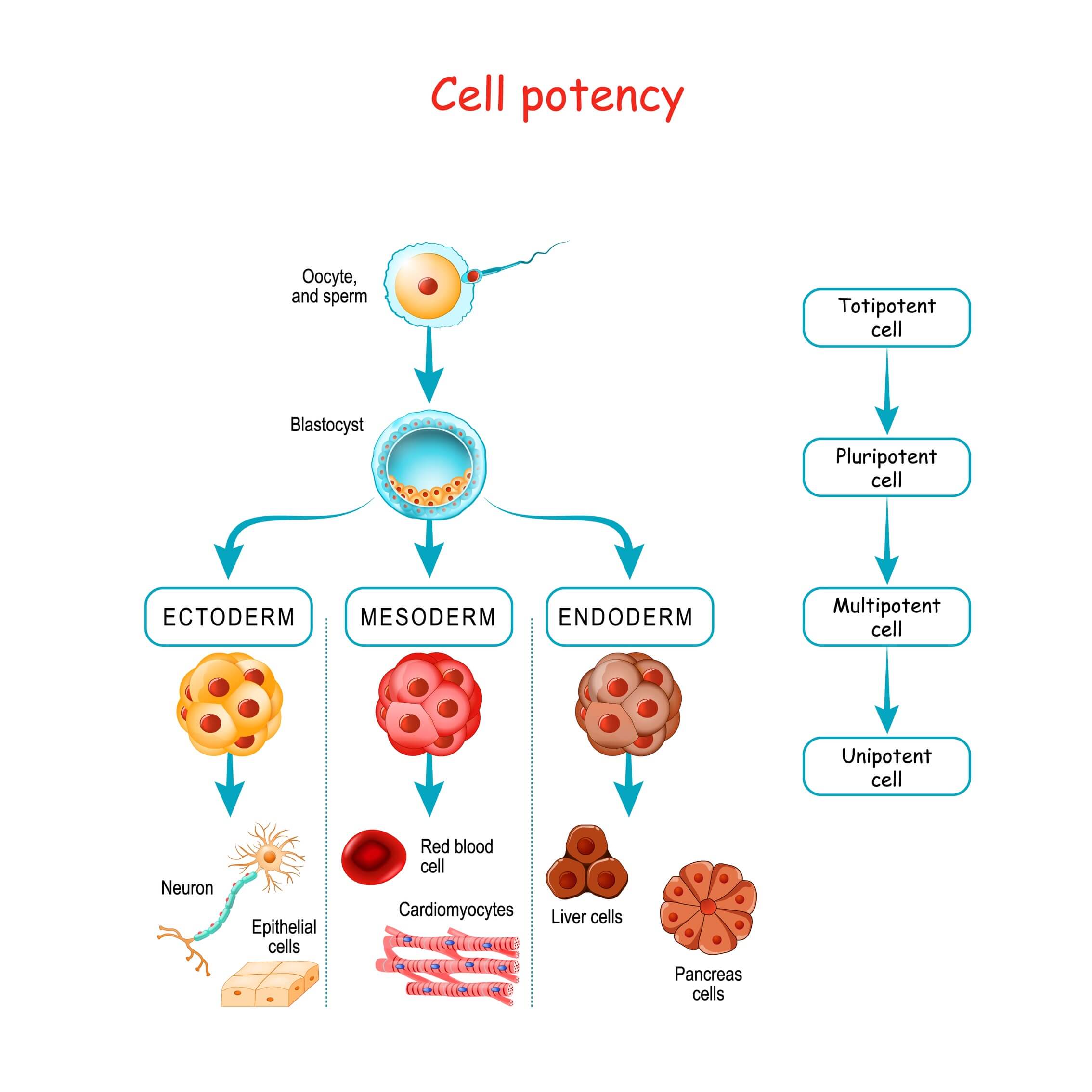
A striking example of this complexity in the context of embryo research lies in the case of parthenogenetic embryos with genomic imprinting defects. While some define an embryo as the product of fertilisation, others consider it as a cluster of human cells with the potential to develop into a viable person. On the other side, proponents contend that the striking similarities hold significant implications for regulatory frameworks, stipulating that integrated SEMs should already be legally and ethically considered as embryo-equivalents.Īs the line between integrated SEMs and natural embryos blurs, the absence of a unified legal definition for an embryo across international borders adds a further layer of complexity. Some argue that integrated SEMs are still far from achieving embryo-equivalent status, emphasising the distinctions that remain between them and natural embryos. Such an outcome could render these models virtually indistinguishable from natural embryos in their potential to develop into living organisms. Yet as research progresses, the possibility of culturing integrated SEMs to term ex utero or until they acquire sentience presents a critical question. Once assembled, mouse ETX embryoids have already passed the window of implantation and therefore cannot implant if transferred into a mouse uterus. This striking resemblance inherently raises ethical concerns. These integrated SEMs exhibit remarkable features, such as a beating heart, ongoing circulation, a neural tube, primordial germ cells, and all extraembryonic lineages.(8,9,10) For instance, mouse ETX models have demonstrated the ability to develop outside the uterus (ex utero), closely resembling their in vivo counterparts at approximately one-third of pregnancy. At the heart of this controversy lies their increasing similarity to natural embryos in terms of cellular morphology, molecular signatures and even developmental potential. Yet, along with their promise, ETX embryoids bring important challenges to fuel broader debate. The ability of SEMs to capture the complexities of early development certainly opens up new horizons for research in regenerative medicine and developmental biology. This is in stark contrast to non-integrated SEMs, including amniotic sac models, gastruloids, axioloids and organoids in general, which capture only specific aspects of embryonic development, leaving critical components absent. Among them are blastoids, which mimic the preimplantation blastocyst, and more recent models that recapitulate the early post-implantation pre-gastrulation embryo, ETX (embryonic–trophoblast–extra-embryonic endoderm) embryoids.(2,3,4,5,6,7) The latter stand out, as they contain all cell types and capture key hallmarks of post-implantation stage embryogenesis up to 14 days post-fertilisation, including bilaminar disk formation, epiblast lumenogenesis, amniogenesis, anterior-posterior symmetry breaking, primordial germ cell specification, primary and secondary yolk sac formation, and extraembryonic mesoderm expansion that defines a chorionic cavity and a connective stalk. Thus far, we have witnessed the emergence of various human integrated SEMs. With their newfound totipotent capabilities, PSCs, whether alone or in conjunction with TSCs or XEN cells, can be reassembled into complete embryo-like structures: integrated stem cell-derived embryo models (SEMs). Armed with a deeper understanding of all these specialised stem cell types, it was merely a matter of time before researchers embraced the challenge of putting the embryo back together again. Notably, with advancements in gene editing and reprogramming methods, scientists have also been able to revert PSCs back to a totipotent state. Similarly, extraembryonic endoderm stem cells (XEN cells) solely give rise to hypoblast-derived lineages. Trophectoderm stem cells (TSCs) represent one such example, differentiating exclusively into trophectoderm-derived lineages. In addition to generating PSCs, scientists have also succeeded in deriving other types of specialised stem cell types from early embryos. During this process, PSCs effectively integrate into the inner cell mass (ICM), but are not incorporated into the trophectoderm.(1) Pluripotency can be demonstrated by the capacity of PSCs to generate a chimeric embryo when introduced into a blastocyst or when aggregated with a morula. Yet unlike totipotent cells, such as zygotes and early blastomeres which can develop into a complete embryo (comprised of both embryonic and extraembryonic tissues), PSCs contribute solely to embryonic lineages, omitting crucial elements that form the placenta and other extraembryonic structures. Pluripotent stem cells (PSC) are so called because of their remarkable ability to differentiate into the three primary germ layers which give rise to the developing embryo.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
